PEARL: A Forced Field Trip or the Experience of a Lifetime?

Katelyn Valla and her biology class took a field trip to Morgan State University’s Patuxent Environmental & Aquatic Research Laboratory to learn about oysters in the Patuxent River.
Biology Field Trip
Program
- Biology (B.A.)
Department
- Biology
Oct. 4, 2018
On Sept. 25, my marine biology class set out on a 2.5-hour trip to Morgan State University’s Patuxent Environmental & Aquatic Research Laboratory (PEARL). It started out as a rough day, waking up early to leave Hood’s campus by 7 a.m. We boarded a big yellow school bus—not exactly what we all had in mind, but it did the trick, I suppose. Did I mention the forecast was calling for rain? The morning started out with about 20 tired and uncomfortable college students forced into a field trip by Professor Ron Albaugh. What happened next was an experience I know I will never forget, and I think most of my classmates would agree.
When we arrived at the site, we were introduced to Richard Lacouture, PEARL’s education coordinator, and Rebekah Borgert, a leader in the oyster hatchery at the laboratory. My group was going to spend the morning on a boat with Richard and Andrew Turner, facilities manager and the boat’s captain, testing the river’s water quality and surveying various species within the river. In the afternoon, we would be with Rebekah, learning how PEARL has been trying to preserve the oyster population in the Patuxent River. It also turns out the weather forecast changed to be a bright sunny day, which might have been a sign of the day to come.
We set out on the boat with Richard, Andrew and Professor Albaugh. While we made our way to the testing area of the river, Richard told us the health of the Chesapeake Bay and the Patuxent River have been steadily declining. Clean-up efforts started about 35 years ago, but there is still a lot of work to be done. At certain times of the year, there is not enough oxygen in the water to sustain life on the bottom, which means many plants and animals living in these ecosystems have been dying. The oyster population (which filters phytoplankton from the water) has been rapidly declining, which means the phytoplankton population has been rising rapidly, because the oysters cannot filter the water to maintain a balance. Attempts to restore the health of these ecosystems include decreasing nutrient runoff by introducing buffer strips that utilize living shorelines. This technique prevents erosion of the shoreline and prevents excess nutrients from entering the lake. The hope is that this will increase the water clarity and allow light to penetrate farther to allow sea grasses to grow in the depths of the waters and increase oxygen levels.
Once we arrived at the testing site, we split into groups to examine the water clarity, phytoplankton levels, salinity (dissolved salt), temperature and dissolved oxygen levels. Utilizing the Secchi disk and underwater light meter, we determined the water clarity was low. We used a fluorometer to acquire the phytoplankton levels, which were high. The phytoplankton decreased with depth, however, because there is insufficient light for photosynthesis at deeper levels.
Finally, we used a YSI multiprobe instrument to measure salinity, temperature and dissolved oxygen levels. The river is entering the fall turnover period, meaning the surface temperatures are beginning to drop. Colder water has a higher density, so it sinks to the bottom. During this turnover period, the river goes through many temperature fluctuations during which the surface water sinks to the bottom as it cools. The salinity levels were quite low, but they increased slightly with depth. One consequence of low salinity levels this year was the absence of jellyfish in the area. The native jellyfish require specific temperatures and salinity levels as part of their reproductive process, and this season never met those requirements because of the excessive amounts of rain this year. The dissolved oxygen levels were also low. After the water quality was tested, a benthic grab was performed, bringing a sample of the sediment at the bottom to the surface. There was no living material since there is not enough oxygen to sustain life at the bottom. The sediment had a fluffy texture, like pudding, and reeked of sulfur due to the presence of anaerobic bacteria producing sulfur as a byproduct at the bottom of the river.
We then headed to the oyster bar and completed an oyster dredge. From the sample, we analyzed how many living oysters and how many box oysters we caught. Box oysters are shells that have remained attached without the animal living inside. The living oysters were also separated according to their shell size. Three inches is the legal limit that allows oysters to be harvested. Anything less must be returned to its natural habitat. After acquiring those results, the oysters and shells were returned. Finally, an otter trawl net was deployed to sample the fish population in the river. The results produced seven different species of fish. After all of those successful results, Andrew steered us back to the dock, and had pizza for lunch, courtesy of our hosts.
After the excitement of our morning, we could not wait to experience what Rebekah had in store for us at the oyster hatchery. First, she gave us a crash course on oysters. She told us they are more than just breathing rocks, and I was amazed at what I learned. Oysters are filter feeders, which means they feed on the algae and microscopic material found in the water column. Each oyster can filter 50 gallons of water per day, and at the height of the river’s health and the oysters’ population, the bay could be filtered in three to four days. Now, in the current conditions, that same process takes an entire year. The bay oysters used to have a lifespan of 10 to 15 years; now, their lifespan is five years. The size of oyster shells has also decreased drastically. Thanks to new diseases and overharvesting, oysters are now much smaller than in the past. Something must be done to preserve the bay’s oyster population before it is lost. The bay’s salinity is currently too low, which means the oyster larvae cannot survive to adulthood. Conservation efforts include decreasing nutrient runoff, adding more oyster shells into the ecosystem, limiting harvesting, and oyster farming.
Rebekah then performed an oyster dissection, showing us exactly how the oyster functions, and its anatomy. We were even able to experience its heartbeat! Our next activity was an oyster grazing experiment, which involved placing oysters in tanks with algae and measuring the remaining algae levels over time. I was amazed at how quickly the oysters could filter the water. Next, we examined various specimens under the microscope, including diatoms, comb jellies, and plankton. Our final activity was venturing to see the oyster hatchery downstairs. We were able to see firsthand the work Dr. Ming Liu is doing with genetic selection in the oysters. The patterns on each of their shells was amazingly distinct and purposeful. There is a bright future for oysters with the proper conservation efforts and care.
This trip to Morgan State University’s PEARL is one I will never forget. What started out as a morning full of dread turned into a day of great weather and eye-opening experiences. It was full of education and newfound wonder. I was amazed at what the results of just water quality testing could predict about the health of the ecosystems within the river and surrounding bay. I was filled with curiosity when I learned what I was missing about the world of oysters. There is so much to learn about our marine ecosystems across the world, including right in our backyard.
PEARL Field Trip

Otter Trawl

Otter Trawl

Oyster Grazing

Left to right: Katelyn Valla, Heidi Gonzalez Ramirez and Rebekah Borgert

Professor Albaugh with a Blue Crab

Oyster Dredge

Oyster Hatchery

Patuxent River

Patuxent River
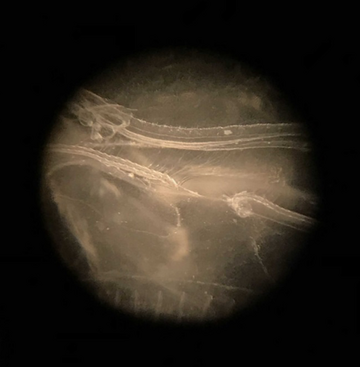
Comb Jelly in Microscope
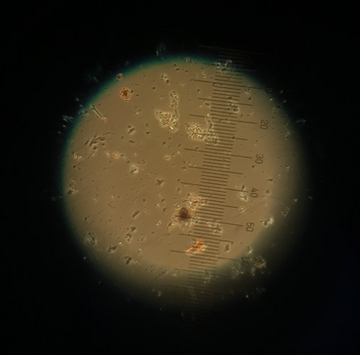
Diatoms in Microscope

Left to right standing: Santana Velasquez, Ron Albaugh, Sara Dickerson, Kyle Scott, Nick Pilny, Heidi Gonzalez Ramirez, Zo Mawi, Gavin Turowski. Front left to right: Katelyn Valla, and Aliyah Buckman

Sara Tievy and Richard Lacouture

Left to right: Kyle Scott, Nick Pilny, Zo Mawi, Heidi Gonzalez Ramirez and Rebekah Borgert

Left to right: Zo Mawi, Heidi Gonzalez Ramirez and Rebekah Borgert

Boat on the Patuxent River

Collecting Data
Are you ready to say Hello?
Choose a Pathway
Information will vary based on program level. Select a path to find the information you're looking for!
